How Do You Know if an Atom Is Stable?
Nuclear Magic Numbers
- Page ID
- 1483
Nuclear Stability is a concept that helps to identify the stability of an isotope. The two main factors that determine nuclear stability are the neutron/proton ratio and the total number of nucleons in the nucleus.
Introduction
A isotope is an element that has same atomic number but different atomic mass compared to the periodic tabular array. Every element has a proton, neutron, and electron. The number of protons is equal to the diminutive number, and the number of electrons is equal the protons, unless information technology is an ion. To determine the number of neutrons in an element you subtract the atomic number from the atomic mass of the element. Atomic mass is represented as (\(A\)) and atomic number is represented as (\(Z\)) and neutrons are represented every bit (\(Northward\)).
\[A=North + Z \characterization{1}\]
The principal factor for determining whether a nucleus is stable is the neutron to proton ratio. Elements with (\(Z<20\)) are lighter and these elements' nuclei and have a ratio of one:1 and adopt to have the same amount of protons and neutrons.
Example \(\PageIndex{1}\): Carbon Isotopes
Carbon has iii isotopes that scientists commonly used: \( \ce{^12C}\), \( \ce{^13C}\), \( \ce{^14C}\). What is the the number of neutron, protons, total nucleons and \(Due north:Z\) ratio for the \( \ce{^12C}\) nuclide?
Solution
For this specific isotope, in that location are 12 total nucleons (\(A\)). From the periodic table, we can see that \(Z\) for carbon (any of the isotopes) is six, therefore \(N=A-Z\) (from Equation \ref{1}):
\[12-6=half dozen \nonumber\]
The N:P ratio therefore is 6:half dozen or a 1:one. In fact 99% of all carbon in the earth is this isotope.
Exercise \(\PageIndex{i}\): Oxygen
Place the number of neutron, protons, total nucleons and N:Z ratio in the \( \ce{^12_8O}\) nuclide?
Elements that take atomic numbers from twenty to 83 are heavy elements, therefore the ratio is dissimilar. The ratio is ane.5:i, the reason for this departure is considering of the repulsive force between protons: the stronger the repulsion strength, the more neutrons are needed to stabilize the nuclei.
Neutrons help to separate the protons from each other in a nucleus and then that they do not experience every bit potent a repulsive forcefulness from other.
Isotope Stability
The graph of stable elements is usually referred to as the Band (or Chugalug) of Stability. The graph consists of a y-centrality labeled neutrons, an x-centrality labeled protons, and a nuclei. At the higher end (upper right) of the ring of stability lies the radionuclides that decay via alpha decay, below is positron emission or electron capture, above is beta emissions and elements beyond the atomic number of 83 are only unstable radioactive elements. Stable nuclei with atomic numbers up to about xx have an neutron:proton ratio of about 1:i (solid line).
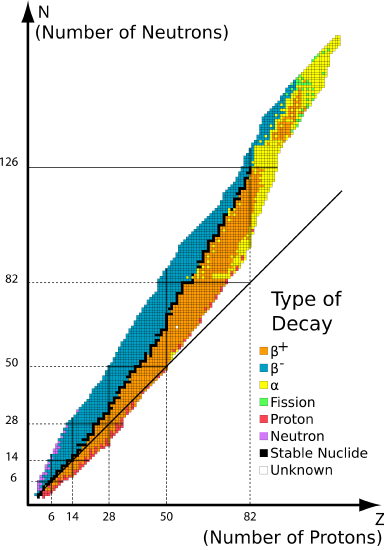
The deviation from the \(N:Z=1\) line on the belt of stability originates from a non-unity \(N:Z\) ratio necessary for full stability of nuclei. That is, more neutrons are required to stabilize the repulsive forces from a fewer number of protons within a nucleus (i.e., \(Northward>Z\)).
The chugalug of stability makes it is easy to determine where the alpha decay, beta decay, and positron emission or electron capture occurs.
- Alpha \(\blastoff\) Decay: Alpha decay is located at the top of the plotted line, considering the alpha decay decreases the mass number of the element to keep the isotope stable. This is accomplished by emitting a alpha particle, which is just a helium (\(\ce{He}\)) nucleus. In this disuse pathway, the unstable isotope's proton number \(P\) is decreased past two and its neutron (\(North\)) number is decreased past 2. The ways that the nucleon number \(A\) decreases by 4 (Equation \ref{1}).
- Beta \(\beta^-\) Disuse: Beta \(\beta^-\) decay accepts protons so it changes the amount of protons and neutrons. the number of protons increase by one and the neutron number decreases by one. This pathway occurs in unstable nuclides that have too many neutrons prevarication above the band of stability (blueish isotopes in Figure \(\PageIndex{i}\)).
- Positron \(\beta^+\) Disuse: Positron \(\beta^+\) emission and electron capture is when the isotope gains more neutrons. Positron emission and electron capture are below the ring of stability because the ratio of the isotope has more protons than neutrons, think of it as there are too few protons for the amount of neutrons and that is why it is below the ring of stability (xanthous isotopes in Figure \(\PageIndex{one}\)).
As with all decay pathways, if the daughter nuclides are not on the Belt, and then subsequent decay pathways will occur until the daughter nuclei are on the Chugalug.
Magic Numbers
The Octet Rule was formulated from the observation that atoms with viii valence electrons were especially stable (and common). A similar state of affairs applies to nuclei regarding the number of neutron and proton numbers that generate stable (not-radioactive) isotopes. These "magic numbers" are natural occurrences in isotopes that are particularly stable. Tabular array 1 list of numbers of protons and neutrons; isotopes that have these numbers occurring in either the proton or neutron are stable. In some cases in that location the isotopes can consist of magic numbers for both protons and neutrons; these would be called double magic numbers. The double numbers merely occur for isotopes that are heavier, because the repulsion of the forces betwixt the protons. The magic numbers are:
- proton: ii, 8, twenty, 28, 50, 82, 114
- neutron: two, 8, xx, 28, l, 82, 126, 184
Also, there is the concept that isotopes consisting a combination of even-even, even-odd, odd-fifty-fifty, and odd-odd are all stable. In that location are more than nuclides that have a combination of even-fifty-fifty than odd-odd. Just like there exist violations to the octet dominion, many isotopes with no magic numbers of nucleons are stable.
| Proton number (Z) | Neutron Number | # of stable Isotopes |
|---|---|---|
| Even | Even | 163 |
| Even | Odd | 53 |
| Odd | Even | 50 |
| Odd | Odd | 4 |
Note
Although rare, iv stable odd-odd nuclides exist: \(\ce{^2_1H}\), \(\ce{^{half dozen}_3Li}\), \(\ce{^{10}_5B}\), \(\ce{^{14}_7N}\)
Unstable or Stable
Here is a simple nautical chart that tin can help you decide is an element is probable stable.
- Summate the total number of nucleons (protons and neutrons) in the nuclide. If the number of nucleons is even, there is a good take chances it is stable.
- Are in that location a magic number of protons or neutrons? 2,8,20,28,50,82,114 (protons), 126 (neutrons), 184 (neutrons) are specially stable in nuclei.
- Calculate the N/Z ratio and use the belt of stability (Figure \(\PageIndex{1}\):) to determine the best way to become from an unstable nucleus to a stable nucleus
Exercise \(\PageIndex{1}\)
Using the above nautical chart country if this isotope is blastoff-emitter, stable, or unstable:
- \(\ce{^{40}_{twenty}Ca}\)
- \(\ce{^{54}_{25}Mn}\)
- \(\ce{^{210}_{84}Po}\)
- Answer
-
Add texts here. Exercise not delete this text first.
Practise \(\PageIndex{2}\)
If the isotope is located above the band of stability what type of radioactivity is it? what if it was beneath?
- Answer
-
Based off the chugalug of stability:
- Stable, because this Ca isotope has xx neutrons, which is on of the magic numbers
- Unstable, considering there is an odd number (25 and 29) of protons and neutrons
- Alpha-emitter, because Z=84, which follows rule/step i on the nautical chart
Exercise \(\PageIndex{three}\)
Carbon is stable
- Reply
-
Carbon is stable
Exercise \(\PageIndex{4}\)
Name one of the isotopes that consist of odd-odd combination in the nuclei?
- Answer
-
Hydrogen-2, Lithium-6, Boron-x, nitrogen-fourteen
References
- Olmsted Three, John and Gregory M William. Chemical science Quaternary Edition. John Wiley and Sons Inc:NJ, 2006.
- Petrucci, Ralph H., William Southward. Harwood, F. Geoffrey Herring, Jeffry D Madura. General Chemistry. Pearson Education Inc: NJ, 2007.
Source: https://chem.libretexts.org/Bookshelves/Physical_and_Theoretical_Chemistry_Textbook_Maps/Supplemental_Modules_%28Physical_and_Theoretical_Chemistry%29/Nuclear_Chemistry/Nuclear_Energetics_and_Stability/Nuclear_Magic_Numbers
0 Response to "How Do You Know if an Atom Is Stable?"
Post a Comment